April 17th 2014
By: Margie King
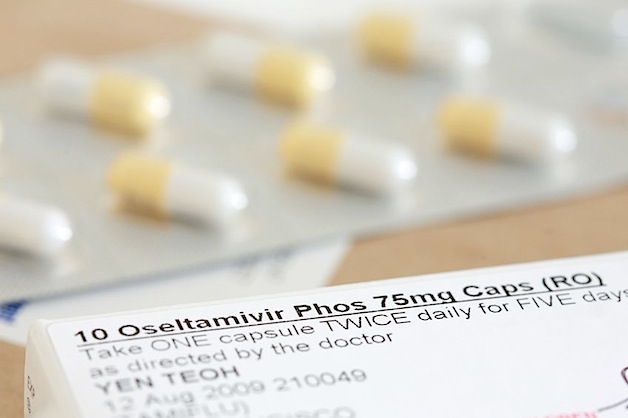
With the outbreak of the H1N1 virus or Swine Flu in April 2009, the worldwide use of Tamiflu skyrocketed. Drug companies and public health agencies claimed it would relieve symptoms and reduce hospital admissions and flu complications like pneumonia, bronchitis, sinusitis, or ear infections.
As fears of an influenza pandemic mounted, the U.S. spent over $1.3 billion to stockpile Tamiflu and Relenza (another antiviral) for use against seasonal and pandemic influenza. The UK government spent almost £424 million to stockpile about 40 million doses of the drugs.
According to a new study, they would have been better off stockpiling a placebo.
Tamiflu (oseltamivir) belongs to a class of drugs known as neuraminidase inhibitors (NIs). Relenza (zanamivir) is also an NI. Tamiflu is used to combat flu in patients as young as 2 weeks of age and Relenza is used in patients aged 5 years and older.
In a scathing review of Tamiflu and Relenza, researchers from The Cochrane Collaboration, an independent, global healthcare research network, are now calling on clinicians and healthcare policy-makers to "urgently revise current recommendations for use of the neuraminidase inhibitors (NIs) for individuals with influenza."
Their findings challenge the historical assumption that NIs are effective in combating influenza. The authors found "no evidence for patients, clinicians or policy-makers to use these drugs to prevent serious outcomes, both in annual influenza and pandemic influenza outbreaks."
The researchers reviewed full internal reports of 20 Tamiflu (Hoffman-LaRoche) and 26 Relenza (GlaxoSmithKline) trials involving more than 24,000 people. This information had previously been withheld for years by the drug companies.
Based on the review, the researchers found the drugs had a "minimal effect on prevention." They found little support for use of NIs as prophylactic agents during influenza epidemics.
According to the study, Tamiflu reduced the term of flu symptoms a meager 14.8 hours from 7 days to 6.3 days compared to placebo. It did not reduce hospitalizations or complications in adults or children.
But Tamiflu did significantly increase the risk of nausea and vomiting in adults (4%) and children (5%). And when it was used in prevention trials risk of headaches, psychiatric disturbances, and renal events increased.
One of the worst side effects may be that Tamiflu stopped some people from producing enough of their own antibodies to naturally fight the flu infection.
Similarly, Relenza reduced the duration of flu symptoms by 14.4 hours from 6.6 days to 6 days in adults compared with a placebo drug. There was no significant effect in children. Relenza also failed to reduce the risk of flu complications or hospitalization.
How could major governments and health organizations recommend and stockpile a drug found to be so useless and even harmful?
The stockpiling was based on recommendations from organizations like the World Health Organization (WHO) and the Centers for Disease Control and Prevention (CDC). The WHO even listed Tamiflu as an essential medicine for the treatment of seriously ill patients or those in higher-risk groups with influenza.
But what were their recommendations based on?
The Cochrane researchers say the original evidence on these drugs presented to government agencies around the world was incomplete. As early as 2009, they tried to verify the safety and effectiveness of neuraminidase inhibitors. But the drug manufacturers refused to provide full access to clinical trial data.
It took years as well as a relentless campaign against the drug companies before they would release the trial data for the drugs. When it finally became public the researchers found that 60% of patient data from randomized, placebo-controlled, phase III treatment trials of Tamiflu had never been published. That included the biggest treatment trial ever undertaken on oseltamivir with over 1400 people of all ages.
This is the first Cochrane review based on all relevant full clinical study reports of a class of drugs integrated by regulatory comments. In other words, they had access to all of the clinical studies submitted to regulatory agencies for approval. They did not rely on trial data as reported in biomedical journals.
In the past these types of clinical study reports have generally remained hidden from public view. Only selectively published trials were available. That has to change according to the authors who said, "Drug approval and use cannot be based on biased or missing information any longer. We risk too much in our population's health and economy."
The findings of this study go far beyond exposing Tamiflu and Relenza. They show clearly that the drug approval system is broken. They emphasize the need to make public all clinical studies by drug companies so they can be independently evaluated.
The researchers called their years-long battle to wrest the relevant studies from GlaxoSmithKline and Hoffman-LaRoche a "small but significant victory for open science."
Dr Fiona Godlee, Editor-in-Chief, The BMJ, said: "This review is the result of many years of struggles to access and use trial data, which was previously unpublished and even hidden from view. It highlights with certainty that future decisions to purchase and use drugs, particularly when on a mass scale, must be based on a complete picture of the evidence, both published and unpublished. We need the full data from clinical trials made available for all drugs in current use."
For information on natural flu remedies read Research Confirms Herbal Medicines Inhibit Influenza .






Leave a comment