The global market for cancer drugs has hit $100 billion in annual sales, and could reach $147 billion by 2018, according to a new report by the IMS Institute for Healthcare Informatics, a unit of drug data provider IMS Health.
This figure does not include discounts or rebates paid to insurers and government programs; IMS says that in the case of cancer drugs, this should not make a big difference the overall figure.
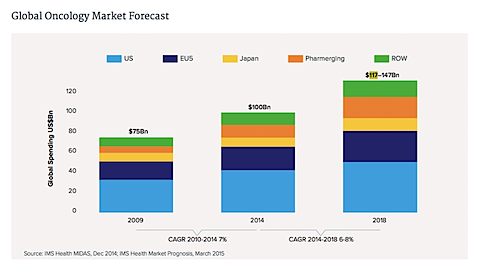
“We’re sitting on the edge of an enormous amount of innovation that is really going to transform the landscape for providers, for patients, and for those companies bringing these products to market,” says Murray Aitken, executive director of the IMS Institute. Below are some highlights from the report.
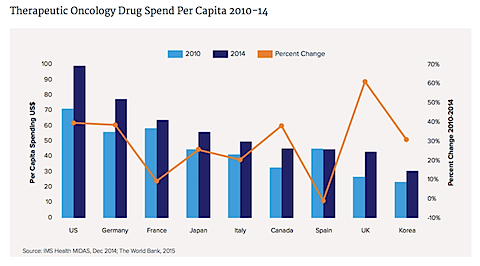
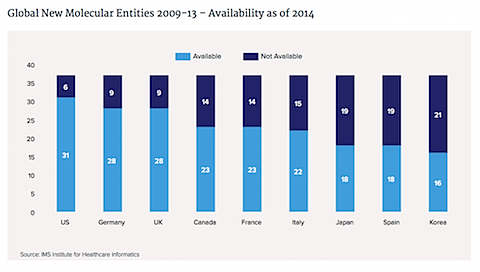
The biggest increase on a percentage basis was in the U.K.; the lowest was in Spain.
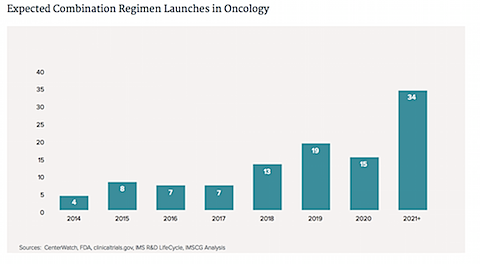
5. Roche is developing the biggest number of combos by a wide margin.
Merck, Bristol-Myers Squibb, and Novartis come next. The figures in blue are for drug combos that include medicines that are already approved made by another company; those in teal include multiple experimental drugs from just a single company; those in purple are testing a new drug with an approved one, both from the same company; those in red test two experimental drugs from different companies.
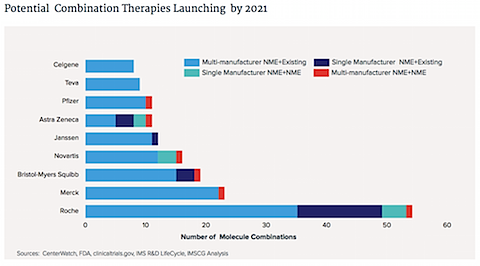
6. More cancer drugs are approved in the U.S. than anywhere else.
Put differently, if a cancer drug is approved anywhere, it is most likely to be approved in the U.S.
7. All these new cancer drugs come at an economic price.
The cost per month for a new cancer drug has increased 40%, or $5,900, over the past decade.
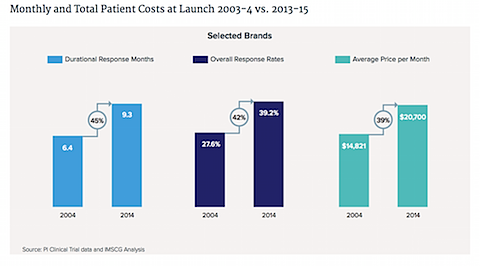
8. More of these costs are being borne directly by patients themselves, not just insurance companies.
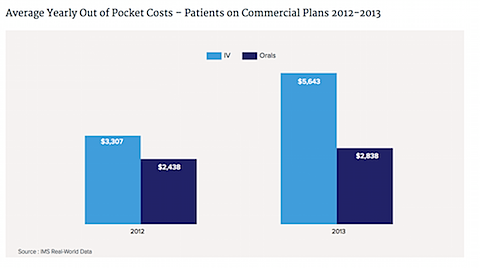
source: http://www.forbes.com/sites/matthewherper/2015/05/05/cancer-drug-sales-approach-100-billion-and-could-increase-50-by-2018/






As there is no drugs for the cancer, the available drugs are becoming more and more useable to reduce the pain and symtomps of the cancer.Such a great help with essay writing at fastessaywriting.net which helps the students to prepare their rresearch paper for the submission.