Restrictions on Food Supplements are Based on Misinformation
FOR IMMEDIATE RELEASE
Orthomolecular Medicine News Service, October 16, 2012
An alert from Europe to the rest of the world
by Gert Schuitemaker, PhD
Introduction: "It can't happen here" qualifies for top placement on the all-time list of famous last words. The United States still has, for now, over-the-counter access to nutritional supplements. But no one who reads newspapers, watches televised news, or leafs through a magazine can miss the preponderance of negative reporting on vitamins. As OMNS continues to counter such misinformation (this issue is the 145th), we take a look at the real "risks" of dietary supplements. Readers may wish to keep in mind what Dr. Abram Hoffer famously said: "All attacks on supplement safety are really attacks on supplement efficacy." If supplements are vilified, they can be made prescription. If they are prescription, costs will go up and access will vanish. - Andrew W. Saul, Editor
(OMNS Oct 16, 2012) A recent study explains that the risk of mortality from taking food supplements is far lower than other risks like smoking, pharmaceutical adverse drug reactions, cancer, and even dying from a lightning strike. [1] This important new information is relevant to recent food regulations in the European Union (EU) that are supposed to make commercially sold food supplements safer. The study shows the belief that food supplements are dangerous is mistaken.
The Codex Alimentarius was established In 1963 by the Food and Agriculture Organization of the United Nations (FAO), the World Health Organization (WHO) and later the World Trade Organization (WTO) as an international standard, with guidelines and codes of practice for the sale of food products, including food supplements.[2] In the natural health community, the Codex is considered a threat to freedom of choice and purchase of food supplements because it stipulates what doses of supplements can be sold and what wording may be used in advertising and packaging.
The Codex has not been adopted by the United States, but within the EU, it was signed into law in 2002 with the adoption of the European Food Supplements Directive. This set of regulations restricts the free choice of consumers when purchasing food supplements. To more fully appreciate this issue, it should be understood that compared to the United States, the EU is highly socialized and regulated. Acceptance of such rigid legislation by policy makers and politicians is easier in Europe than on the other side of the Atlantic. But giant food corporations are lobbying for similar limitations in the USA. Thus, the Codex Alimentarius and the EU legislation are considered a likely template for exporting this type of food regulation to the rest in the world.
This type of food legislation is designed to protect every citizen of the EU from suspected risks, even those imagined to be related to taking food supplements. Thus, if a dietary supplement does not have "scientific evidence that it is not harmful," it is treated as harmful until proven otherwise. . On first thought, many of us would expect that the government has a moral obligation and an implied mandate to research such risks and impose such precautionary laws. However, this paralyzing "dangerous until proven safe" logic recently drove the EU committee in charge to deny claims that water treats dehydration and prunes treat constipation, because there was not enough scientific evidence to make these claims! [1]
Threat to freedom of health
The implementation of the European Food Supplements Directive is imminent. As of December 14, 2012, health claims made on food supplements must be authorized by the European Food Safety Authority (EFSA), based on a very rigid and restrictive set of rules. Of the 4000 claims submitted so far, only about 220 have been accepted. For example, the regulations forbid the use of terms such as 'energy' for coenzyme Q10, 'antioxidant' for quercetine, and 'probioticals' for probioticals on supplement labels. The reason is that the law considers these terms to be unfounded claims of health benefits.
This EU legislation is in opposition to the wishes of consumers who want to take responsibility for their own health. Citizens worldwide fighting for freedom of choice should take note, because the template for global implementation has been set in motion with nothing to stop its assault on your health freedom. In response to this insidious threat, the Alliance for Natural Health International compiled a chart that quantifies the risk of mortality from various causes within the EU.[1]
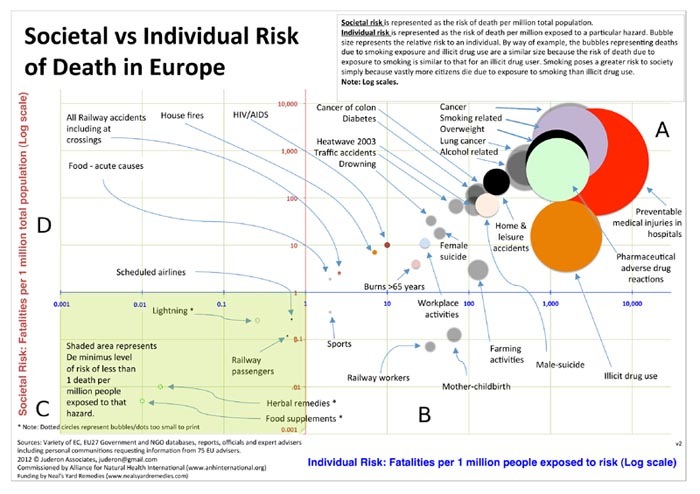
http://www.orthomolecular.org/resources/omns/v08n31-figure1-lg.jpg
Figure 1. Risk of death from various causes in the EU. First, note the position of the bubbles in the quadrants. The X-axis (horizontal) indicates the risk of mortality for an individual (per million people) when the individual is exposed to the risk. The Y-axis (vertical) indicates the overall risk of mortality per million EU residents. A.The upper right quadrant shows mortality risks that apply to a relatively large proportion of the population, for example cancer or preventable medical injury, and that are also relatively large for individuals when exposed. B. The bottom right quadrant shows that a relatively small proportion of the total EU-population dies from the risk, for example railway work, but an individual has a relatively large risk of mortality from exposure. C. The bottom left quadrant shows a relatively small risk of mortality for the overall EU-population, and that an individual also has a small risk of mortality from exposure. D. The upper left quadrant shows a theoretical risk which is relatively large for the overall population, but is small for an individual when exposed. In reality this cannot occur because if individual risk of death is small, then overall the risk must also be small. The size of each bubble represents the relative risk for individual exposure. Note: the log scale is used to allow the data to be meaningfully presented on one graph, but this implies that the differences in risk are exponentially greater than shown by the bubble positions. Figure adapted from ANHI [1].
Results of the ANHI risk study
Result 1. Smoking and illicit drug use have equally large bubbles (upper right quadrant). This means an equal and relatively large risk of mortality from smoking and drug use for the individual smoker and individual drug user (about 2,500 per million [1:400]). But the overall risk for drug use is much lower because fewer people use illicit drugs than smoke (risk from illicit drug use: ~10 per 1 million EU inhabitants [1: 100,000]; from smoking: ~1,000 per million [1 : 1,000]).
Result 2. Lightning and use of food supplements (lower left quadrant) both represent an extremely small risk. The risk of mortality from food supplement use by individuals is 1 in 100 million, and for being struck by lightning is 80 in 100 million. So the risk of mortality as a result of a food supplement use is 80 times smaller than from lightning.
Result 3. Comparison of the use of food supplements (lower left quadrant) vs. preventable medical injuries in hospitals (upper right quadrant). The relative size of the bubbles shows that the risk of mortality from food supplements is much lower than the risk of mortality from preventable medical injuries in hospitals. Reading from the X-axis, it is apparent that the risk of mortality to individuals from food supplements is extremely small (1 in 100 million), but the risk of mortality from preventable medical injuries in hospitals is thousands of times greater (5,000 per million or 0.5%). Reading from the Y-axis, it is apparent that the overall risk of mortality in the EU from food supplements is even lower (6 in 1 billion or 1 in 170 million) and from preventable injuries during a stay in the hospital about 700 per million [1 in 1400].
Result 4. - Comparison of risk for individual (represented by bubble size and the location on the X-axis):
Use of food supplements Compared to preventable medical injuries in hospitals 1:351,220
Compared to smoking 1:173,000
Compared to cancer 1:173,000
Compared to pharmaceutical adverse drug reactions 1:123,125
Compared to lightning 1:26
Use of herbal remedies Compared to preventable medical injuries in hospitals 1:206,600
Conclusion
Taking your daily food supplement in the EU is one of your safest daily activities. Even getting fatally struck by lightning is a bigger risk. The risk of dying from preventable medical injuries in hospitals is 350,000 times greater. The European authorities meant to impose precautionary legislation to protect the EU citizen against the risks they imagined, but in fact their prejudice favoring large food corporations is shown by the risk chart. Food supplements are very, very safe. The Codex Alimentarius is considered by many as an imminent threat to the freedom to take the food supplements of citizens of the US. Unfortunately, like the food legislation in the EU, it is currently being considered as a template for legislation worldwide [3]. This most definitely includes America.
(Dr. Gert Schuitemaker trained as a pharmacist and then completed his PhD in medicine at University of Maastricht. He is the founder of the Ortho Institute in the Netherlands, which publishes Orthomoleculair magazine for health professionals and Fit mit Voeding ("Fit With Nutrition") for the public. Dr. Schuitemaker has published several books and more than 300 articles.)
For further information:
The Alliance for Natural Health International is a non-governmental organization promoting natural and sustainable approaches to healthcare. ANHl campaigns across a wide range of healthcare fields, including the use of herbal products and essential nutrients in adequate doses. http://www.anh-europe.org and http://www.anh-usa.org
References:
1. http://www.anh-europe.org/news/anh-exclusive-lightning-more-dangerous-than-herbs-or-vitamins.
2. http://www.codexalimentarius.org/.
3. Schwitters B. Health Claims Censored. The case against the European Health Claim Regulation. De Facto Publications, 2012.
Nutritional Medicine is Orthomolecular Medicine
Orthomolecular medicine uses safe, effective nutritional therapy to fight illness. For more information: http://www.orthomolecular.org
Find a Doctor
To locate an orthomolecular physician near you: http://orthomolecular.org/resources/omns/v06n09.shtml
The peer-reviewed Orthomolecular Medicine News Service is a non-profit and non-commercial informational resource.
Editorial Review Board:
Ian Brighthope, M.D. (Australia)
Ralph K. Campbell, M.D. (USA)
Carolyn Dean, M.D., N.D. (USA)
Damien Downing, M.D. (United Kingdom)
Dean Elledge, D.D.S., M.S. (USA)
Michael Ellis, M.D. (Australia)
Martin P. Gallagher, M.D., D.C. (USA)
Michael Gonzalez, D.Sc., Ph.D. (Puerto Rico)
William B. Grant, Ph.D. (USA)
Steve Hickey, Ph.D. (United Kingdom)
Michael Janson, M.D. (USA)
Robert E. Jenkins, D.C. (USA)
Bo H. Jonsson, M.D., Ph.D. (Sweden)
Thomas Levy, M.D., J.D. (USA)
Stuart Lindsey, Pharm.D. (USA)
Jorge R. Miranda-Massari, Pharm.D. (Puerto Rico)
Karin Munsterhjelm-Ahumada, M.D. (Finland)
Erik Paterson, M.D. (Canada)
W. Todd Penberthy, Ph.D. (USA)
Gert E. Schuitemaker, Ph.D. (Netherlands)
Robert G. Smith, Ph.D. (USA)
Jagan Nathan Vamanan, M.D. (India)
Andrew W. Saul, Ph.D. (USA), Editor and contact person. Email: omns@orthomolecular.org Readers may write in with their comments and questions for consideration for publication and as topic suggestions. However, OMNS is unable to respond to individual emails.
To Subscribe at no charge: http://www.orthomolecular.org/subscribe.html